Everything you need to know about endometriosis
 Endometriosis is a common cause of chronic pelvic pain and female infertility. Statistically, endometriosis affects 6-10% of women of reproductive age, 50-60% of women and teenage girls with pelvic pain, and up to 50% of women with infertility. In this blog we will discuss the most important facts you need to know about endometriosis.
Endometriosis is a common cause of chronic pelvic pain and female infertility. Statistically, endometriosis affects 6-10% of women of reproductive age, 50-60% of women and teenage girls with pelvic pain, and up to 50% of women with infertility. In this blog we will discuss the most important facts you need to know about endometriosis.
What is endometriosis?
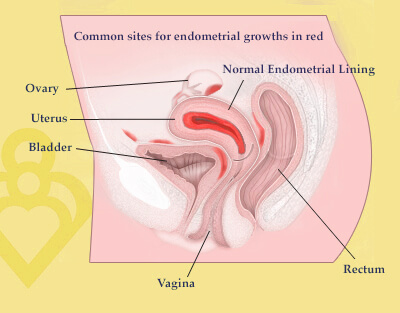
Endometriosis is a common disease in which cells from the normal uterine lining – called endometrium – attach to structures outside the uterine cavity, implanting and growing and responding to hormonal changes. Common places to find endometriosis include the pelvic lining (peritoneum), ovaries, external bladder, and bowel as well as the posterior ligaments which support the uterus within the pelvis and the tissue between the vagina and rectum. In rare cases, endometriosis can occur in more unusual locations such as the diaphragm, lungs, the membranes surrounding the lungs and the heart, or the liver.
Endometrial cells trigger an inflammatory response outside the uterus
While the exact cause of endometriosis is still a mystery, it is thought to occur by the mechanism of “retrograde menses”. In other words, normal live endometrial cells “backwash” through open fallopian tubes into the pelvis during a woman’s period. These cells are otherwise normal, and as such, depend on estrogen for growth, just like normal endometrium in its normal location inside the uterus. The cells grow and shed on these surfaces during the menstrual cycle and, in doing so, cause an inflammatory response.
In some women this inflammation is very intense and is eventually associated with adhesion formation, anatomical distortion, scarring, and infiltration by aberrant nerve fibers. This can lead to pain and infertility. Ovarian cysts filled with endometriosis are called “endometriomas”. They are thought to start as small encapsulated superficial lesions that subsequently expand. Endometriosis at distant anatomical sites is thought to be caused by spread from peritoneal lesions.
Endometriosis affects women differently
Although most women have retrograde flow during menses, not all women develop endometriosis. Furthermore, not all women with endometriosis have intense inflammation, progression of lesions, pain, and infertility. The true prevalence of the disease among asymptomatic women is unknown.
It is possible that all women have some endometriosis but only a minority has problems as a consequence. The reasons behind this wide variability are among the greatest mysteries in medicine and have been the subject of investigation since the disease was first described in 1898. There appears to be some degree of genetic susceptibility, likely related to the immune system, but other factors appear to play a role as well, including certain combinations of lifestyle and/or environmental exposures.
Accurately diagnosing endometriosis can only be done surgically
There is no reliable, non-invasive way to accurately diagnose endometriosis. In current clinical practice, a surgical procedure called laparoscopy is required to visualize and, if necessary, biopsy the lesions. An ultrasound exam can help identify ovarian endometriomas, but the majority of lesions occurs on the peritoneal surfaces and cannot be seen on ultrasound.
At the time of surgery, endometriosis is staged according to the number and size of the lesions: Stage I (minimal), II (mild), III (moderate) and IV (severe). Staging is useful in determining disease burden and management but, interestingly, does not correlate at all to the severity of pain or predict response to therapies for pain or infertility. Among asymptomatic women who present with infertility, the proportion that have endometriosis at laparoscopy ranges from 9-50% (average around 30%) and the majority (70% or more) have small amounts of disease, typically Stage I-II.
The majority of studies in this field show that infertile women with Stages I-II endometriosis have virtually the same chances of successful conception with fertility therapy as infertile women without endometriosis. Since the therapeutic benefit of laparoscopy to improve fertility among women without symptoms appears to be minimal at best, routine diagnostic laparoscopy is usually not necessary.
Endometriosis can cause infertility
How is endometriosis linked to infertility? Scientists still are not sure of the exact relationship between endometriosis and infertility, although several theories have been proposed. Obviously, scarring around the ovary and fallopian tube may prevent the normal release of an egg and pick-up from the ovary to the fallopian tube. In some women, the normal fluids that promote cell-to-cell communication, called cytokines, interleukins, prostaglandins and proteases, may be present in abnormal amounts which then promotes inflammation or hinders the events that must occur in the proper sequence for normal fertilization and implantation. Other theories suggest egg and embryo quality may be impaired or that the normal endometrium in the proper location may be less receptive or not express the right receptors at the right time.
Managing endometriosis symptoms depends on a woman’s fertility goal
What is the best way to manage endometriosis? Among women who present with chronic pelvic pain, the likelihood that endometriosis is present is 30-80%. Management strategies depend on the fertility goals of the patient. Studies show that women with mild endometriosis (Stage I-II) don’t benefit from medical or surgical management. But women with Stages III-IV (moderate to severe) endometriosis have somewhat lower chances of conception and often need more organized and aggressive fertility treatment to ultimately succeed. In vitro fertilization has become a powerful and effective tool for women with endometriosis as it maximizes the chance of successful conception.
For women struggling with pain and infertility, treatment options and the chances of successful conception have never been better than they are today. Even though many questions about the true causes and pathological mechanisms of endometriosis remain unanswered, the majority of women suffering with endometriosis are able to lead healthy lives and complete their families as desired. Contact our fertility specialists to discuss your chances of pregnancy with endometriosis.



